Nuclear Physics Formulas
Nuclear Physics is a study of atomic nuclei. In other words, it's all about the components and structure of the nucleus. If you ever need help on Nuclear Physics Concepts we are always there for you. In fact, you can enhance your level of understanding regarding the topics lying by using Nuclear Physics Formulas. The Formula Sheet of Nuclear Physics existing includes concepts like Atomic Mass Unit, Size of Nuclei, Mass Defect, and Nuclear Binding Energy, etc. For more concepts and related guidance have a glance at our Physics Formulas and clear all your queries.
Nuclear Physics Formulas Sheet & Tables
1. Nuclear force
A strong force acts among the nucleons in the nucleus. This force is called nuclear force. The range of this force is of the order of 10-14 m. Nuclear force does not depend on charge, depends on spin.
2. Size of nucleus
Nucleus is assumed spherical. Its radius and mass number are related as
R = Ro A1/3 = 1.25 × 10-15 A1/3 m
3. Atomic mass unit (amu)
One amu is equal to \(\left(\frac{1}{12}\right)\) of the mass of one atom of 6C12.
1 amu = 1u = 1.66 × 10-27 kg
Energy of 1 amu = 931.5 MeV
4. Mass defect and nuclear binding energy
In reality the mass of the nucleus is always less than the total mass of its components. The difference is called mass defect. The energy equivalent to this mass defect is called nuclear binding energy.
(a) Mass defect = sum of masses of all nucleons – mass of the nucleus >
Δm = (Zmp + Nmn) – M
= [Zmp + (A – Z)mn – M]
(b) Packing fraction = \(\frac{\text { Mass defect }}{\text { Mass number }}\)
or f = \(\frac{\Delta \mathrm{m}}{\mathrm{A}}=\left(\frac{\mathrm{M}-\mathrm{A}}{\mathrm{A}}\right)\)
(c) Nuclear binding energy = (Δm)c2 = Δm (a.m.u) × 931.5 MeV
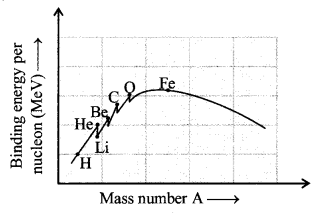
(d) Binding energy per nucleon
= \(\frac{\text { Nuclear binding energy }}{\text { Mass number }}\)
= \(\frac{(\Delta \mathrm{m}) \mathrm{c}^{2}}{\mathrm{A}}\)
\(\overline { B.E } \) = \(\frac{\left[\mathrm{Zm}_{\mathrm{p}}+(\mathrm{A}-\mathrm{Z}) \mathrm{m}_{\mathrm{n}}-\mathrm{M}\right] \mathrm{c}^{2}}{\mathrm{A}}\)
Binding energy curve:
5. Nuclear reactions
Nuclear reactions take place when the nucleus of an atom is bombarded with high energy protons, neutrons, α-particles, deuterons, γ-rays etc.
Following conservation laws are obeyed in the nuclear reactions:
- Mass-energy conservation law.
- Linear momentum conservation law.
- Angular momentum conservation law.
- Charge conservation law.
- Nucleon conservation law.
Q-value- The equivalent energy corresponding to the net loss of mass in a reaction is called ‘Q’ value.
As in ZXA → Z1XA1 + Z2XA2 + Q
Q = (Mx – MY1 – MY2)c2
= (A1Eb1 + A2Eb2 – AEb)
Where Eb is binding energy per nucleon.
6. Neutrons
Neutron was discovered by Chadwick in 1932. Neutron is a neutral particle. Its mass is slightly greater than that of proton. Mass of neutron is 1.675 × 10-27 kg.
7. Nuclear fission
The process in which a heavy nucleus breaks-up into two nearly equal nuclei and a large amount of energy is released is called nuclear fission
92U235 + 0n1 → (92U236) → 56Ba141 + 36Kr92 + 30n1 + Q
Q ≈ 200 MeV per fission
Atomic bombs based on the principle of nuclear fission of uncontrolled chain reaction.
8. Controlled chain reaction and atomic reactor
In controlled chain reaction the rate of fissions is controlled. Therefore, this j process is slow. This process is used for the production of electricity in an I atomic reactor. Main parts of an atomic reactor are:
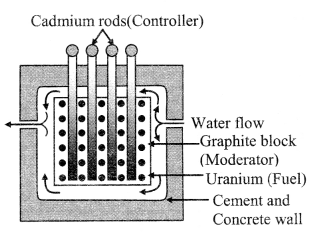
- Nuclear fuel as U235, Pu239
- Moderator as D2O (Heavy water), graphite.
- Coolant as water, air, CO2, liquid Na.
- Controller as cadmium.
- Shield as thick concrete wall or thick lead sheet
9. Nuclear fusion
When two light nuclei combine to form a heavy nucleus, the process is called nuclear fusion. This process is possible only at very high temperatures ~107 K. Energy produced in sun and stars is due nuclear fusion.
41H1 → 2He4 + 2β+ + 2v + Q
Q ≈ 24.7 MeV per cycle.
Need instant help while solving the physics concepts formulas you can always look up to Onlinecalculator.guru a trusted portal for all your doubts.