Solution And Colligative Properties Formulas
The formula sheet of Solution And Colligative Properties are prevailing here for making you feel comfortable to solve all the equations of the solutions given in the exams. You can exactly learn all about Solution And Colligative Properties concept by using the important Formulas list presented on this page. Go through the below sections and have a look at the Solution And Colligative Properties Formulas tables and cheatsheet to grasp the concept easily.
Solve your chemistry problems fastly and efficiently taking the help of Chemistry Formulas and learn about the Concepts without much effort.
Cheat Sheet for Solution And Colligative Properties Formulas
Don't miss the chance of learning Solution And Colligative Properties concept basics by using the formula sheet provided here. Not anymore of feeling low when calculating the Solution And Colligative Properties concept equations wrong, because we have come with the Solution And Colligative Properties Formulas list for you. Just make use of the Solution And Colligative Properties Formula sheet & revise the concept easily & thoroughly.
1. Molarity = \(\frac{\text { No. of moles of solute }}{\text { Volume of solution (litre) }}\)
& Molarity = \(\frac{\text { No.of moles of solute }}{\text { mass of solvent }(\mathrm{kg})}\)
m = \(\frac{1000 \mathrm{M}}{1000 \mathrm{d}-\mathrm{MM}_{\mathrm{B}}}\) & Molarity ∝ \(\frac{1}{\text { temp. }}\)
XA = \(\frac{\mathbf{n}_{\mathbf{A}}}{\mathbf{n}_{\mathbf{A}}+\mathbf{n}_{\mathbf{B}}}\) and XB = \(\frac{\mathrm{n}_{\mathrm{B}}}{\mathrm{n}_{\mathrm{A}}+\mathrm{n}_{\mathrm{B}}}\)
Mole percent = mass fraction × 100
& Mass fraction of A = \(\frac{\mathbf{W}_{\mathbf{A}}}{\mathbf{w}_{\mathbf{A}}+\mathbf{w}_{\mathbf{B}}}\)
ppm = \(\frac{\text { Mass of solute }}{\text { Mass of solution }} \times 10^{6}\)
= \(\frac{\text { wt. of solute }}{\text { wt. of solute }+\text { wt. of water }} \times 10^{6}\)
Normality = \(\frac{\text { gram equivalent of solute }}{\text { Volume of solute (liters) }}\)
& N = \(\frac{\mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{E} \times \mathrm{V}}\)
2. Raoult’s Law:
P = pA + pB = p0A XA + p0BXB
= (1 – XB)p0A + p0B XB
= (p0B – p0A)XB + p0A
\(\frac{P_{0}-P_{S}}{P_{0}}=\frac{n}{n+N}\)
& \(\frac{P_{0}-P_{S}}{P_{0}}=\frac{w \cdot M}{W \cdot m}\), when n << N
3. Ideal Solutions:
ΔHmix = 0 & ΔVmix = 0
4. Non-Ideal solutions:
ΔHmix ≠ 0 & ΔVmix ≠ 0
5. Types of Non-Ideal Solutions:
(a) Non-ideal solutions showing positive deviations
pA > p0A XA, & pB > p0BXB, & PT > pA + pB
(b) Non-ideal solutions showing negative deviations:
pA < p0A XA, & pB < p0BXB, & PT < pA + pB
6. π = \(\frac{n}{V}\)RT = CRT
& \(\frac{p_{A}^{0}-p_{A}}{p_{A}^{0}}=X_{B}=\frac{n}{n+N}\)
& ΔTb = Kb × m \(\frac{\mathrm{K}_{\mathrm{b}} \times \mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{M}_{\mathrm{B}} \times \mathrm{W}_{\mathrm{A}}}\)
ΔTf = Kf × m \(\frac{\mathrm{K}_{\mathrm{f}} \times \mathrm{W}_{\mathrm{B}} \times 1000}{\mathrm{M}_{\mathrm{B}} \times \mathrm{W}_{\mathrm{A}}}\)
& Kb = \(\frac{\mathrm{RT}_{\mathrm{b}}^{2}}{1000 \ell_{\mathrm{v}}}\)
& Kf = \(\frac{\mathrm{RT}_{\mathrm{f}}^{2}}{1000 \ell_{\mathrm{f}}}\)
Colligative properties ∝ Number of particles
∝ Number of molecules (in case of non electrolytes)
∝ Number of ions (In case of electrolytes)
∝ Number of moles of solute
∝ Mole fraction of solute
7. i = \(\frac{\text { Normal molar mass }}{\text { Observed molar mass }}\)
= \(\frac{\text { Observed colligative property }}{\text { Normal colligative property }}\)
i = \(\frac{\text { Observed osmotic pressure }}{\text { Normal osmotic pressure }}\)
= \(\frac{\text { Actual number of particles }}{\text { No. of particles for ionisation }}\)
degree of association
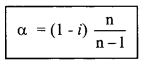
& degree of dissociation
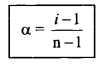
Get a Complete List of Chemistry Formulas from our website Onlinecalculator.guru and refer to the respective formula sheet to apply them while solving related problems to attain accurate solutions.